 Gender identity, including in trans people, appears to have a physical basis and a biological underpinning, and the Endocrine Society has stated that numerous features suggest trans people’s genders are a matter of likely prenatal development:
Gender identity, including in trans people, appears to have a physical basis and a biological underpinning, and the Endocrine Society has stated that numerous features suggest trans people’s genders are a matter of likely prenatal development:
Such evidence stems from scientific studies suggesting that: 1) attempts to change gender identity in intersex patients to match external genitalia or chromosomes are typically unsuccessful; 2) identical twins (who share the exact same genetic background) are more likely to both experience transgender identity as compared to fraternal (non-identical) twins; 3) among individuals with female chromosomes (XX), rates of male gender identity are higher for those exposed to higher levels of androgens in utero relative to those without such exposure, and male (XY)-chromosome individuals with complete androgen insensitivity syndrome typically have female gender identity; and 4) there are associations of certain brain scan or staining patterns with gender identity rather than external genitalia or chromosomes.
The developmental origin of gender identity has led researchers to look for distinct genetic features that may play a role in the development of a gender identity contrary to assigned sex. One area of focus is genes related to sex hormones, as differences in the functioning of sex hormones may affect the degree of prenatal masculinization of the brain, potentially altering gender identity even as the rest of the body develops normally (Foreman et al., 2019). Hare et al. (2009) reported the promising result that among 112 trans women, a greater number of repeats of the CAG sequence were present in the androgen receptor (AR) gene compared to cisgender men. The authors note that this longer length results in androgens such as testosterone being less effective at activating the androgen receptor, possibly resulting in reduced brain masculinization prenatally.
However, despite the differences in prevalence between trans women and cis men in the study, there was a great deal of overlap: 44.6% of the studied trans women did not have the “long” AR variant potentially causing reduced androgen signaling, while 47.6% of the cis men did have this variant. Additionally, a later study (Ujike et al., 2009) did not replicate this result, finding no significant differences in CAG repeats in the AR between trans women and cis men:
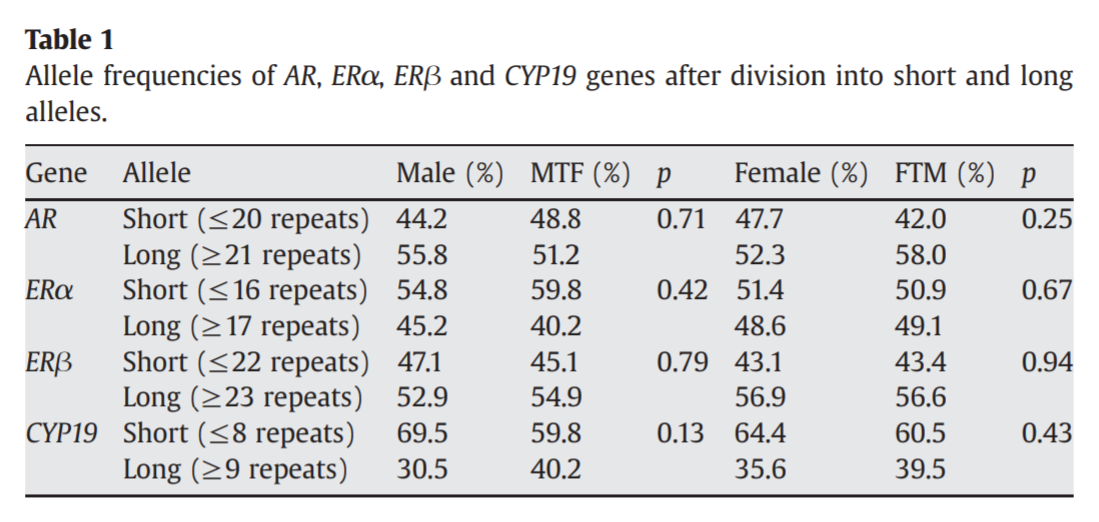
So: Do trans women tend to have a variant of the AR gene that reduces androgen signaling, or do they not? A recent meta-analysis seeks to resolve this uncertainty.
D’Andrea et al. (2020) posit that “small sample sizes in most studies might have contributed to inconclusive results”, and proceed to perform a systematic review of five studies that included information on CAG repeats in trans women’s AR genes. Examining data from a total of 795 trans women and 1,355 cis men, the authors found that “the length of CAG repeat sequences of AR gene was significantly longer in the trans woman group” (p = 0.005). Nevertheless, as with Hare et al., this variant alone is not a reliable indicator of transness:
…the gene polymorphism investigated in the present study is relatively common, suggesting that the revealed association would not reflect a direct independent causal link. It is likely, indeed, that variants in different sex hormone signaling genes involved in brain sexual differentiation, together with other factors, could influence the development of gender identity/incongruence.
In other words: this is not some kind of “trans gene” that can be used to distinguish a trans woman from a cis man just by looking at this one sequence. Instead, the developmental aspects of gender identity may be influenced by many genes related to sex hormones, or interactions between combinations of these genes. Foreman et al. (2019) did not find a difference in AR CAG repeats between trans women and cis men, but four combinations of gene variants were found to be overrepresented in trans women compared to cis men, and three of those combinations involved the long repeat length variant of the AR. The study also identified a number of other gene variants that could alter the functioning of sex hormones in trans women:
Although long CAG repeats in AR alone may not have an independent effect on the development of gender dysphoria, this AR polymorphism may interact with other genes to increase the likelihood of being transgender. … Similarly, long repeats in ERβ have been associated with decreased ERβ signaling, potentially reducing the influence of ERβ on the defeminization of the male brain. In combination, both genotypes appear to have additive effects on the development of gender dysphoria. … Interaction analysis also identified the specific combination of SRD5A2 and CYP17 polymorphism, the former associated with reduced levels of DHT, whereas the latter is known to increase sex steroid precursor production. It seems plausible that together, these polymorphisms may increase the production of precursor steroids and testosterone, but not of DHT, the more potent androgen form.
The authors are careful to highlight that “although discordance rates for gender dysphoria suggest that genetics plays a role, it is not the sole determinant of gender identity.” Additionally, there is the possibility that genetic studies utilizing much larger sample sizes may reveal that these findings of overrepresented gene variants in trans women were false positives – and it may be difficult to acquire genome data from the much larger number of trans women required to offer reasonable assurance that these results are not spurious. Nonetheless, if these are indeed true positive findings, they may provide a valuable piece of the puzzle on the nature, origin, and development of gender dysphoria. ■



